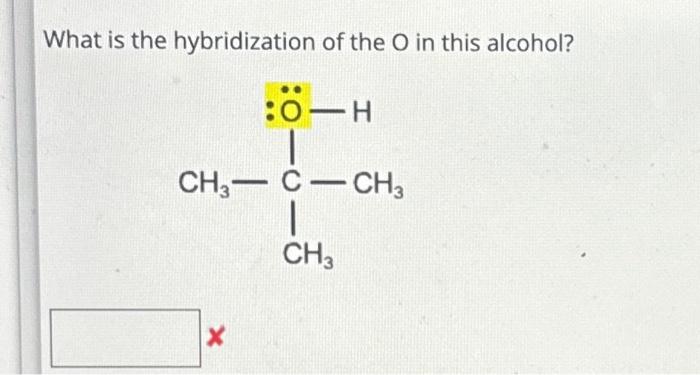
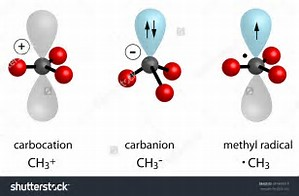
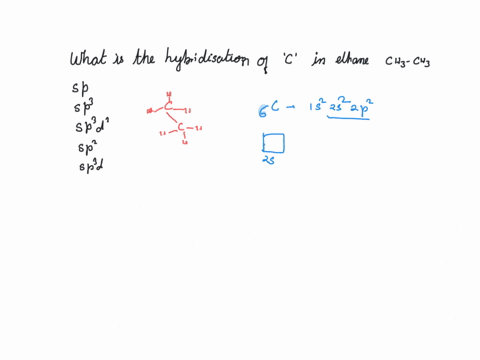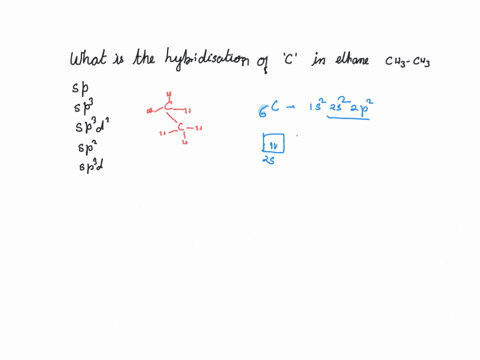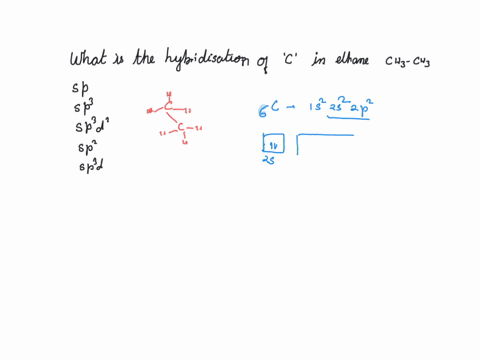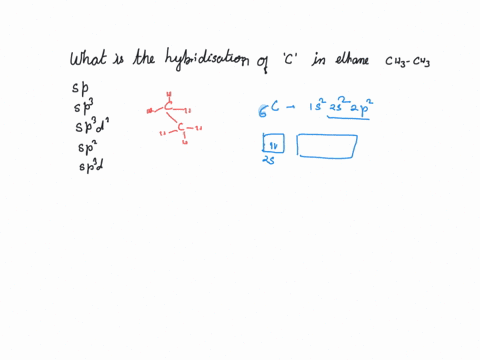What is the hybridization of the carbon atom in each of the following CH_3^+, CH_3^-, CH_3? | Homework.Study.com
Hybridization of central atoms in the moleculesN(CH3)3 and N(SIH3) respectively are(1) sp2 and sp2(2) sp3 and sp3(3) sp2 and sp34) sp3 and sp2
10.What is the hybridization of Oxygen molecule in the following : 1. CH3 O CH=CH2 2. CH3COCH3 [Propanone]

The structure and hybridisation of Si(CH3)4 isa)bent, spb)trigonal, sp2c)octahedral, sp3dd)tetrahedral, sp3Correct answer is option 'D'. Can you explain this answer? - EduRev NEET Question


![Solved] The hybridization of the central carbon in CH3C≡N and Solved] The hybridization of the central carbon in CH3C≡N and](https://storage.googleapis.com/tb-img/production/20/10/F1_Utkarsha_Madhu_20.10.20_D3.png)
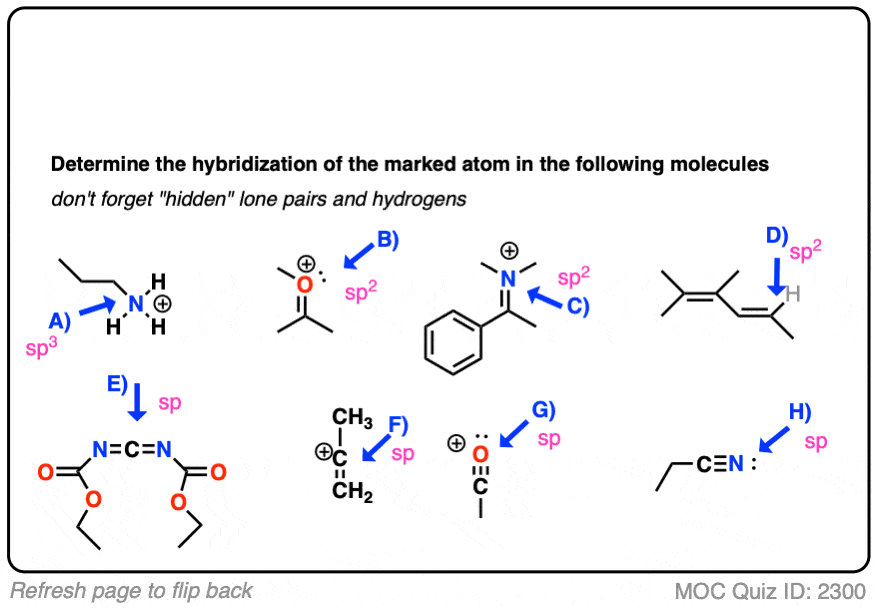


![ANSWERED] What is the hybridization of the N in this amine? CH3-N-CH₂ - Kunduz ANSWERED] What is the hybridization of the N in this amine? CH3-N-CH₂ - Kunduz](https://media.kunduz.com/media/sug-question/raw/76146470-1659792437.8720496.jpeg)