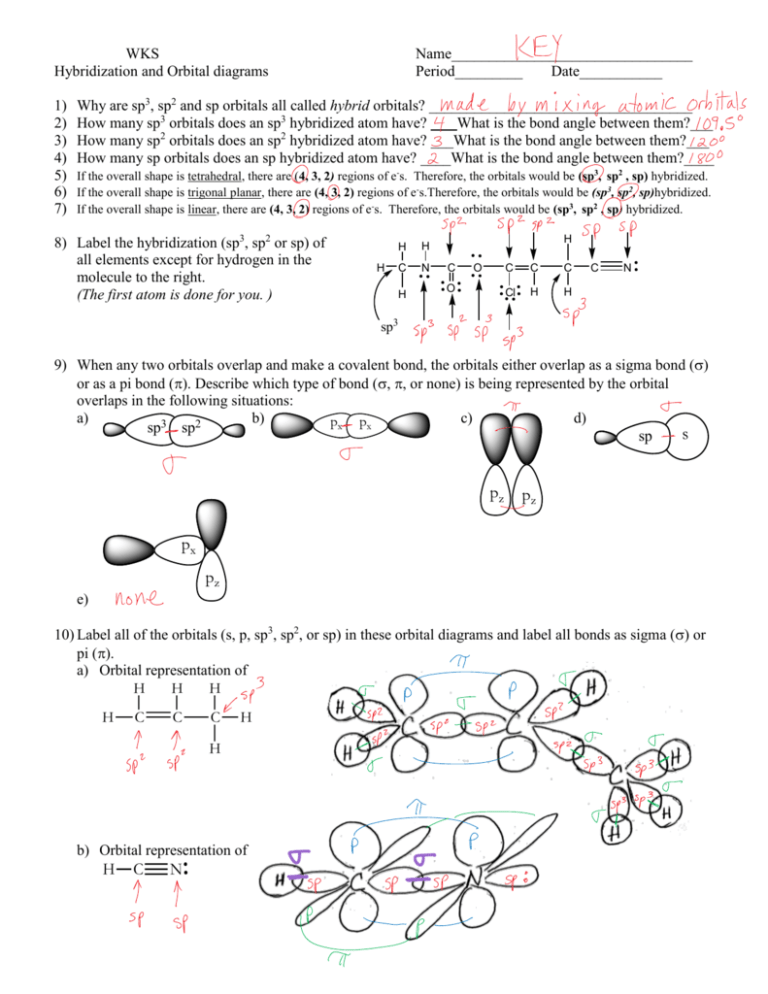
Account for the hybridization at each carbon atom in the mentioned molecule. Chloroprene(used to make neoprene, a synthetic rubber) | Homework.Study.com

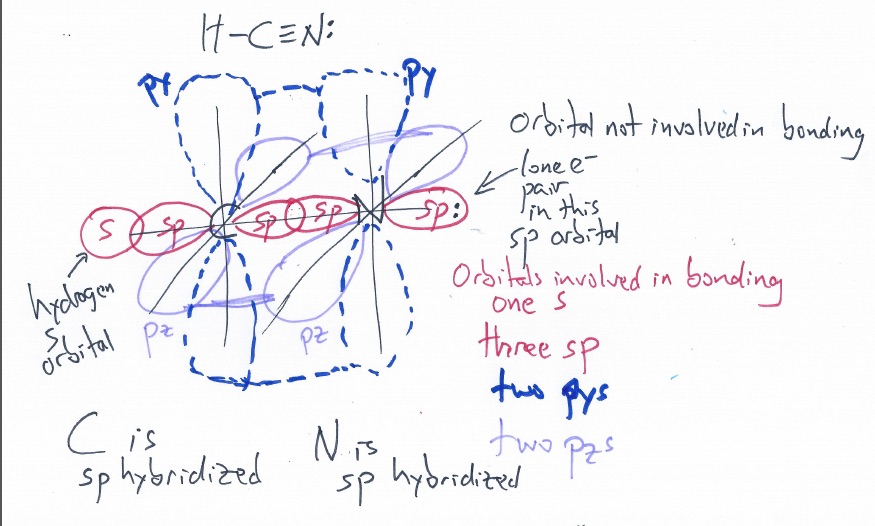
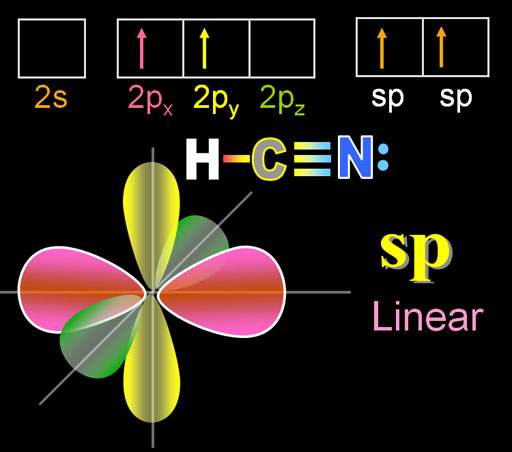
sp hybridization on nitrogen/nitrogen hybridization/HCN hybridization,orbital overlap diagram of hcn - YouTube

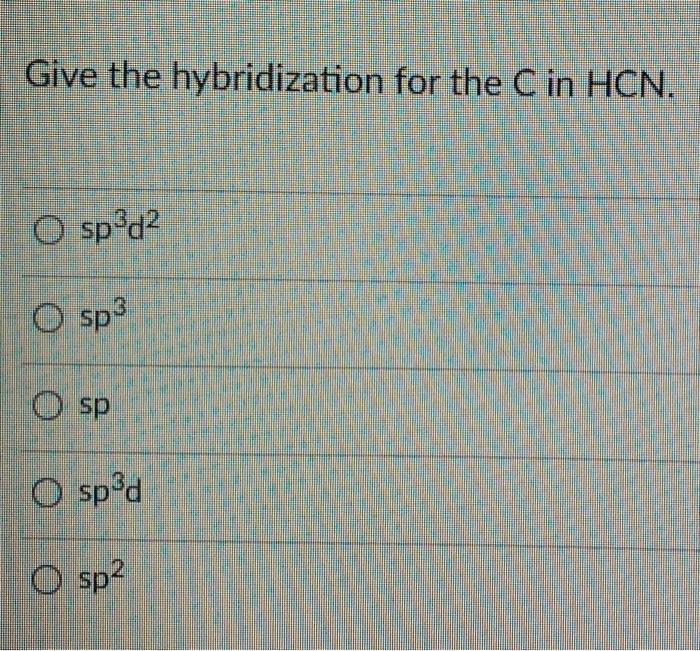
SOLVED: What type of hybrid orbitals are present in HCN? How many sigma and pi bonds are there? (2 marks)