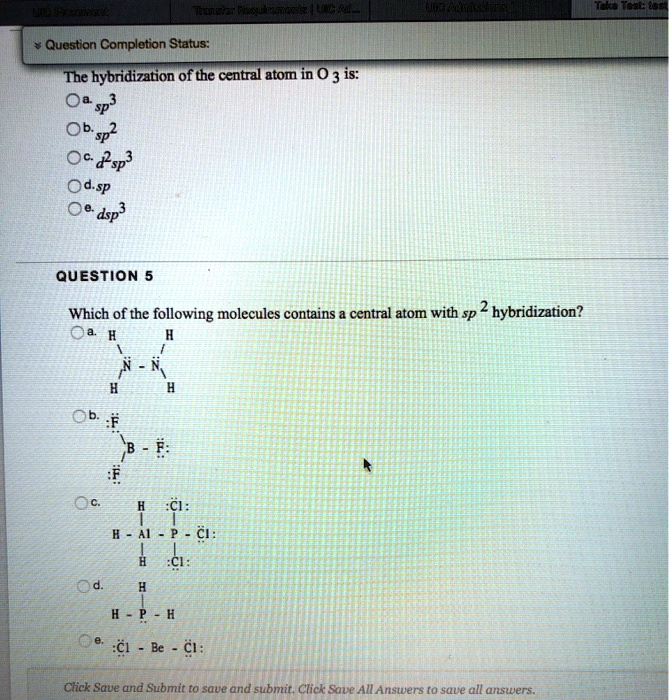
SOLVED: Q4L hybridization of the central atom in O3 is: dsp Which of the following molecules contains a central atom with sp2 hybridization? H2O Cl2 CO2
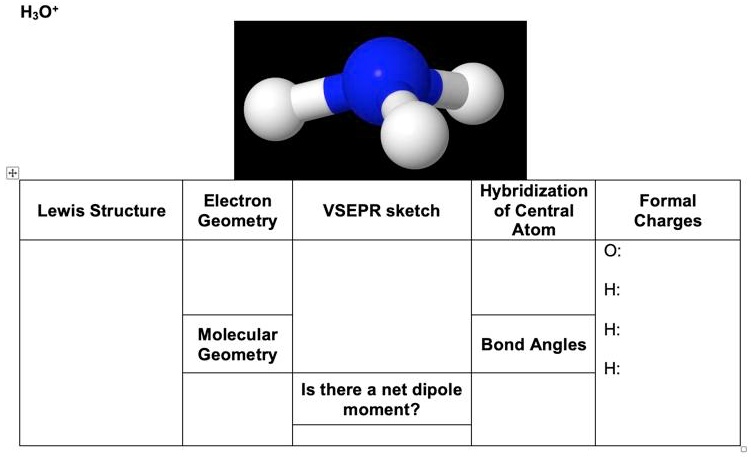
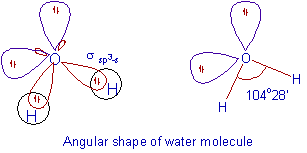
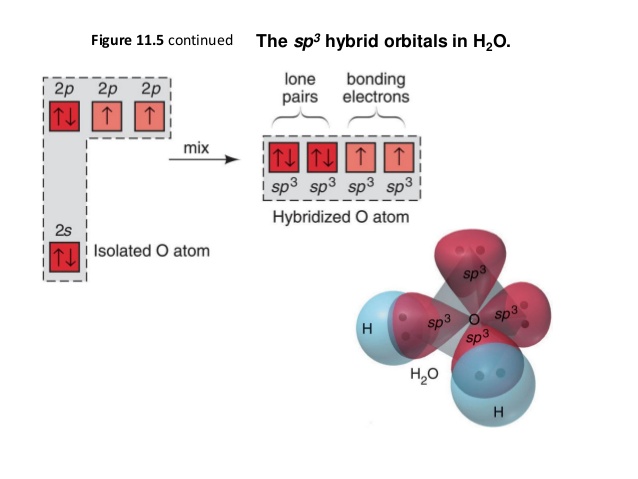
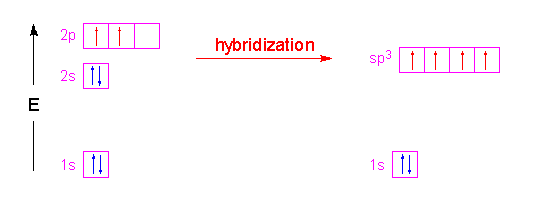
SOLVED: HzO Lewis Structure and Electron Geometry Hybridization of Central Atom Formal Charges VSEPR Sketch Molecular Bond Angles and Geometry H2O: Is there a net dipole moment?

H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs
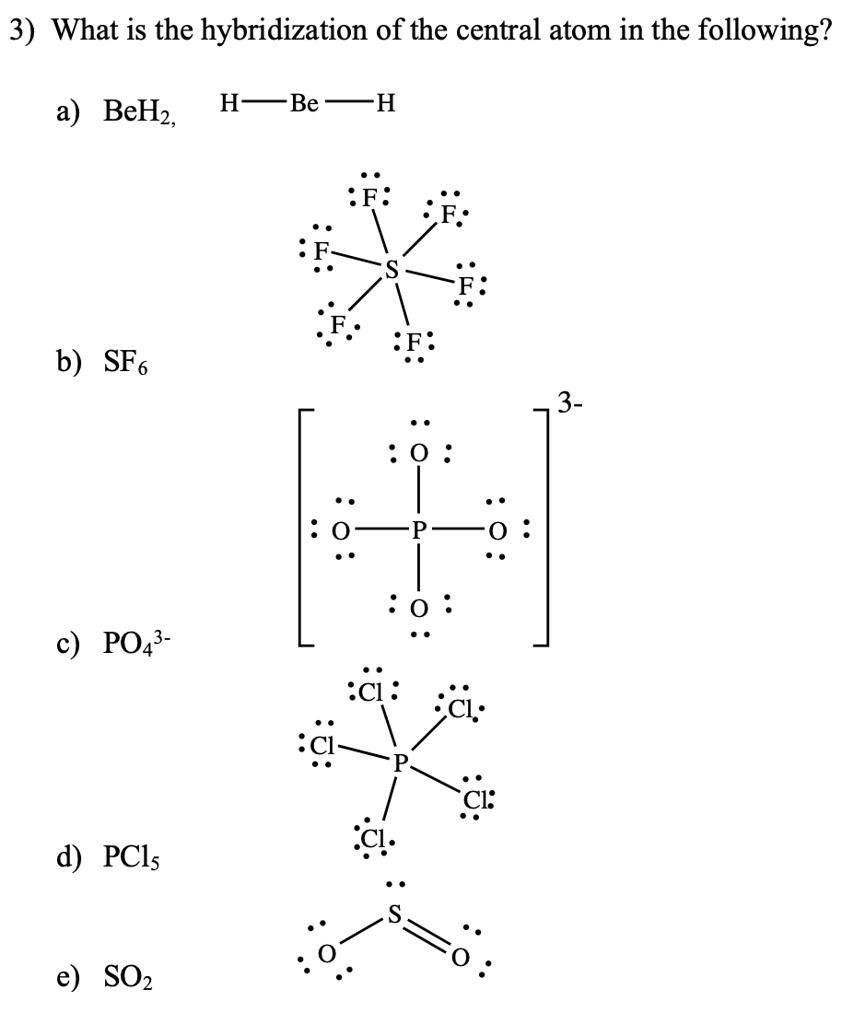
SOLVED: What is the hybridization of the central atom in the following? H2O a) BeH2, b) SF6 c) PO3^- d) PCl5 e) SO2

55. The hybridization of the central atom will change when 1) NHz combines with H+ 2) H,BOcombines with OH- 3) NHz forms NH, 4) H2O combines with H allowed. Use by non