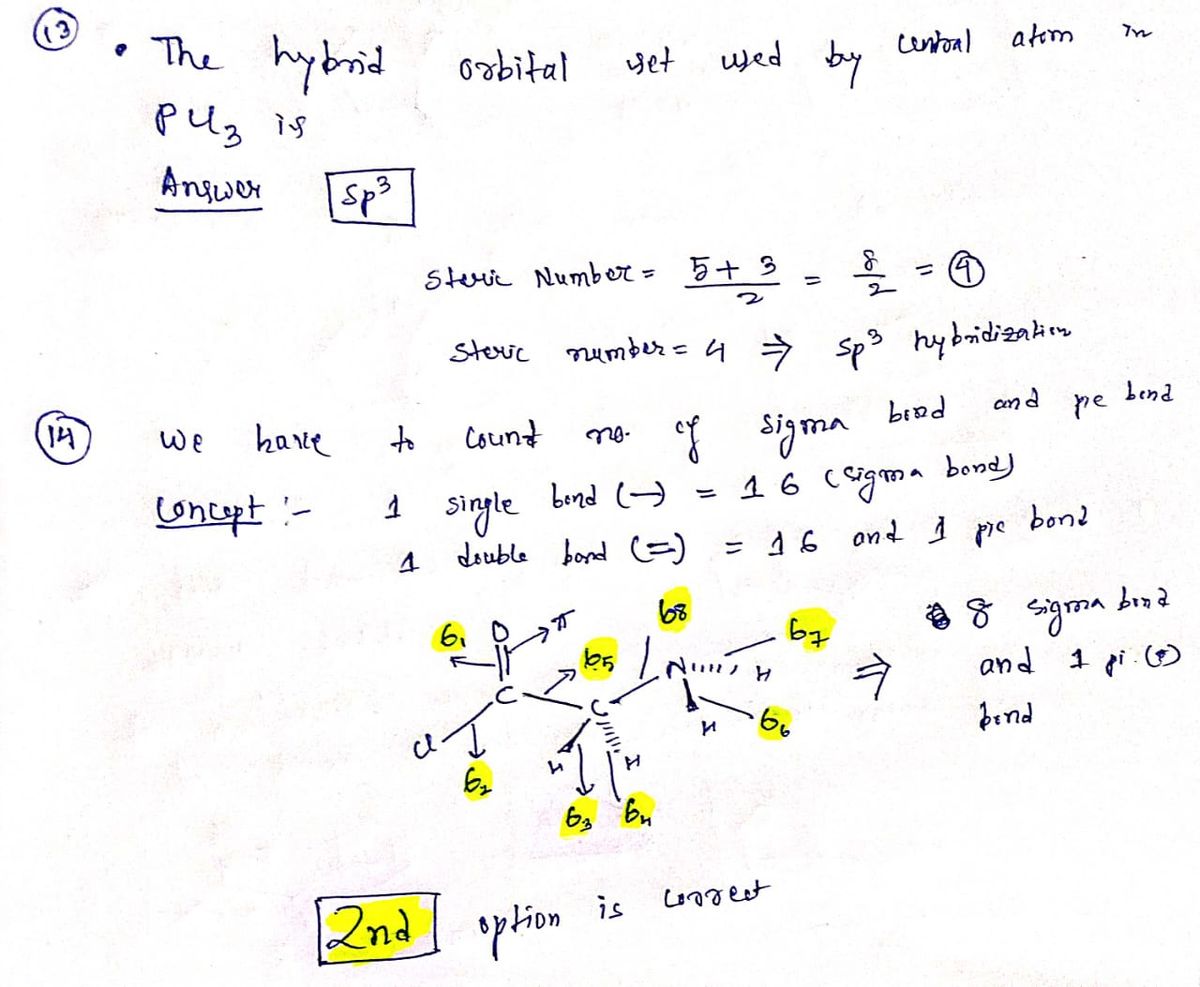
SOLVED: Draw the Lewis structure for BrO4-. What is the hybridization of the central atom (2p orbitals)? sp3 12. Determine the hybridization at each of the three atoms (C, O, E) from
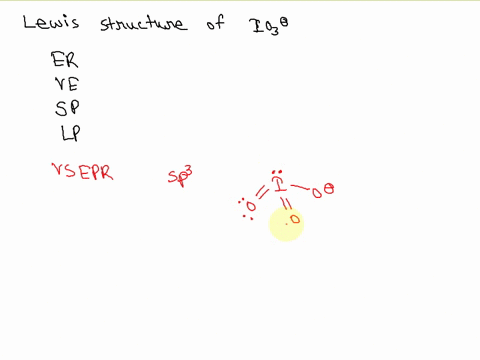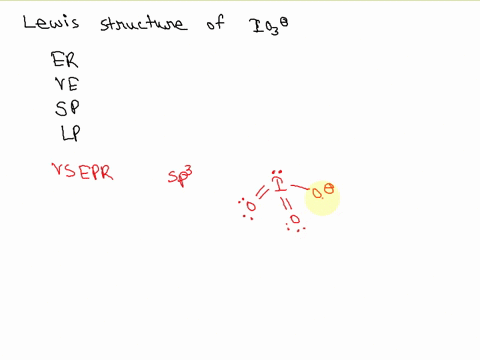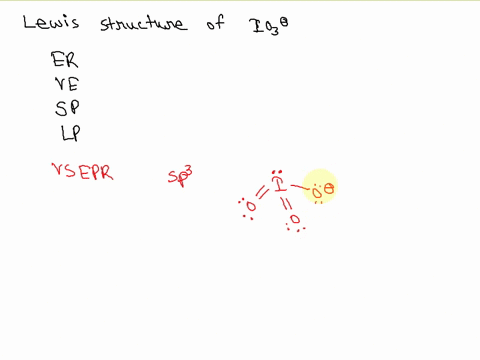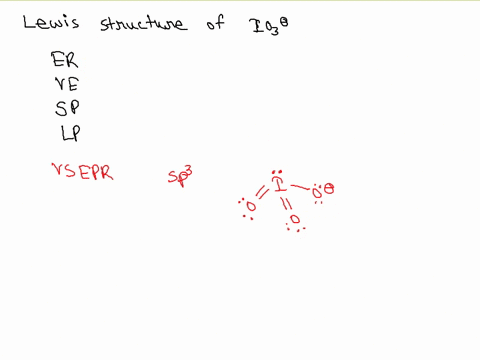
SOLVED: Draw the Lewis structure of IO3- that obeys the octet rule. Calculate the electrons required (ER), valence electrons (VE), shared pairs (SP), and lone pairs (LP). ER: VE: SP: LP:

In the Lewis structure of the iodate ion, IO3-, that satisfies the octet rule, what is the formal charge on the central iodine atom? | Homework.Study.com

SOLVED: In the IO3- ion, iodine is the central atom. Based on the Lewis structure of IO3- that obeys to the Octet rule, what is the formal charge on the iodine atom?
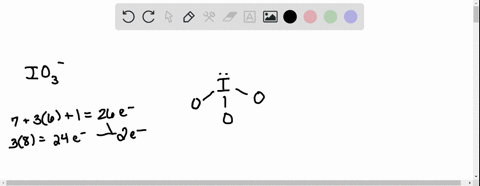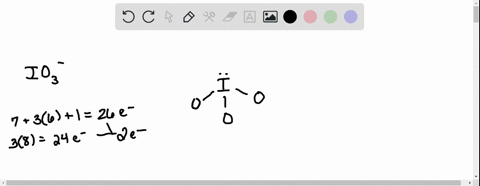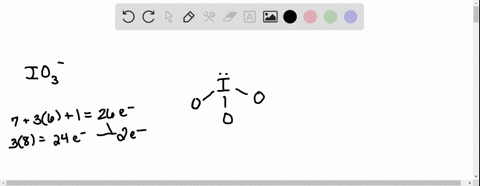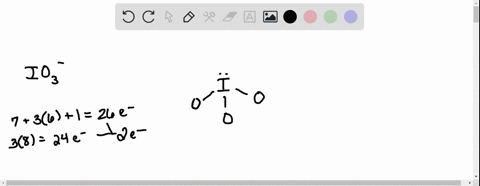
SOLVED: According to the principles of VSEPR theory applied on IO3- ion: a) Draw the most favorable Lewis structure with minimal formal charges (all O atoms are bonded to the central atom). Show all your work. b) Indicate the formal charges of all atoms. c ...

Why the bond angle of XeO3 is 103 - Chemistry - Chemical Bonding and Molecular Structure - 12894693 | Meritnation.com

Deduce the Lewis formula for the given species. Indicate the number of electron groups and the electronic and molecular or ionic geometries. IO3- | Homework.Study.com
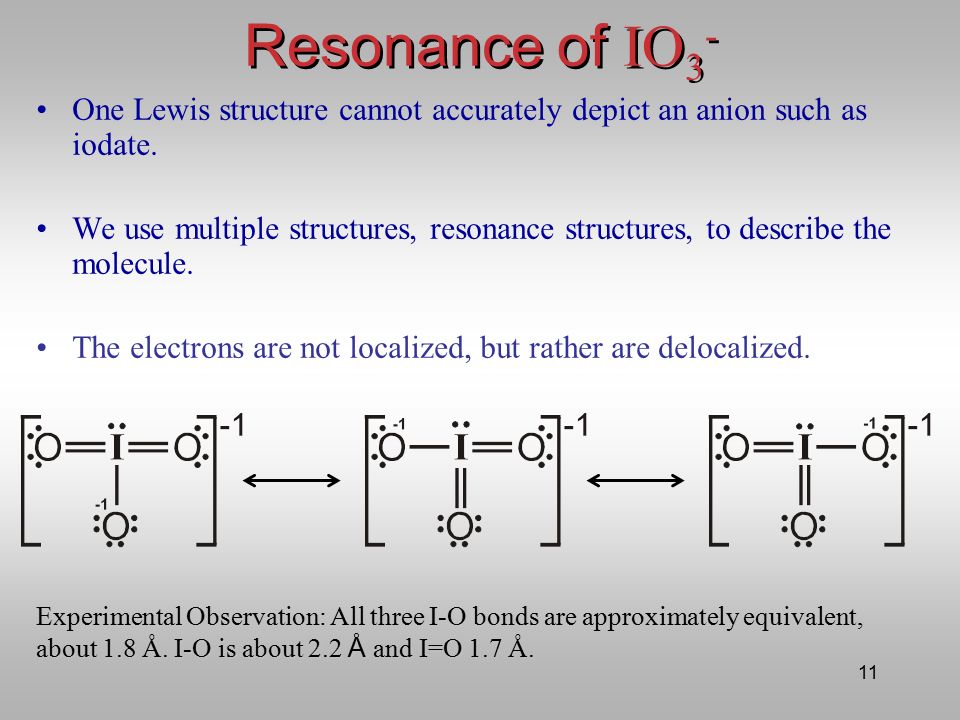
The Structure and Bonding of IO3- An example of the use of Lewis Structures and VSEPR Theory Lecturer: Dr. Andreas Lemmerer. - ppt video online download

BiO(IO3): A New Polar Iodate that Exhibits an Aurivillius-Type (Bi2O2)2+ Layer and a Large SHG Response | Journal of the American Chemical Society


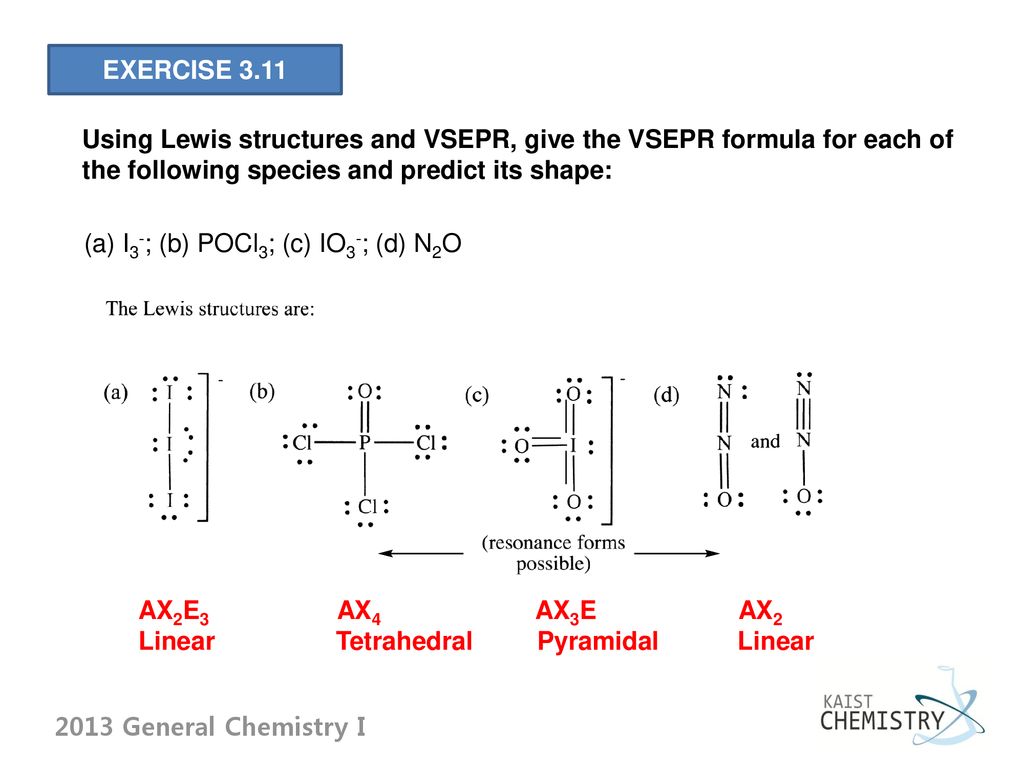

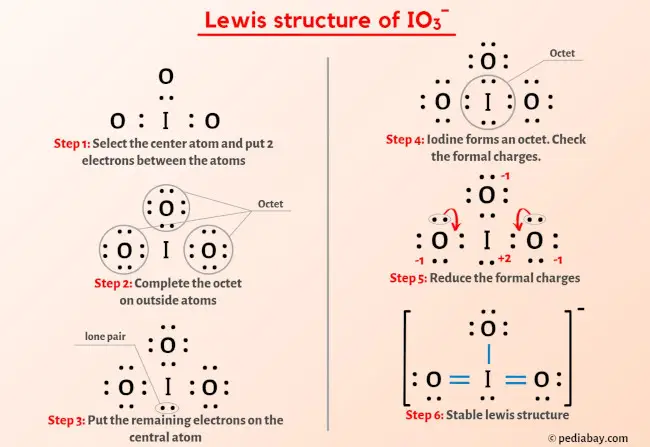





![hybridization process for charged molecule. example [IO3]- - YouTube hybridization process for charged molecule. example [IO3]- - YouTube](https://i.ytimg.com/vi/s39QIsfZZlI/maxresdefault.jpg)