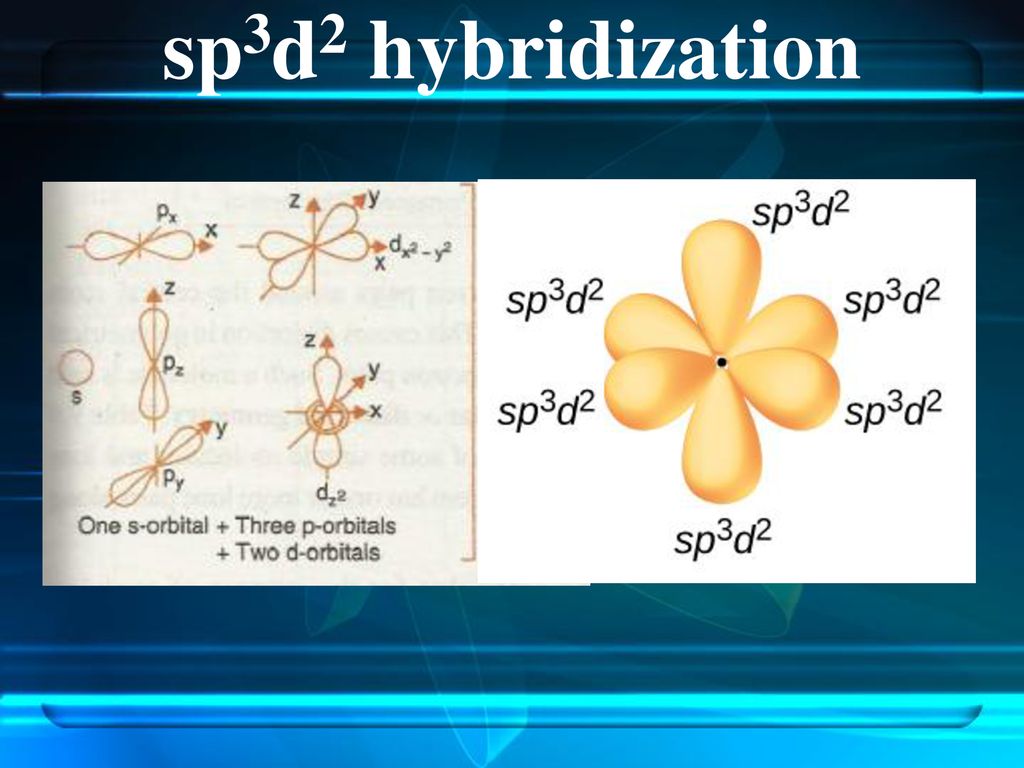
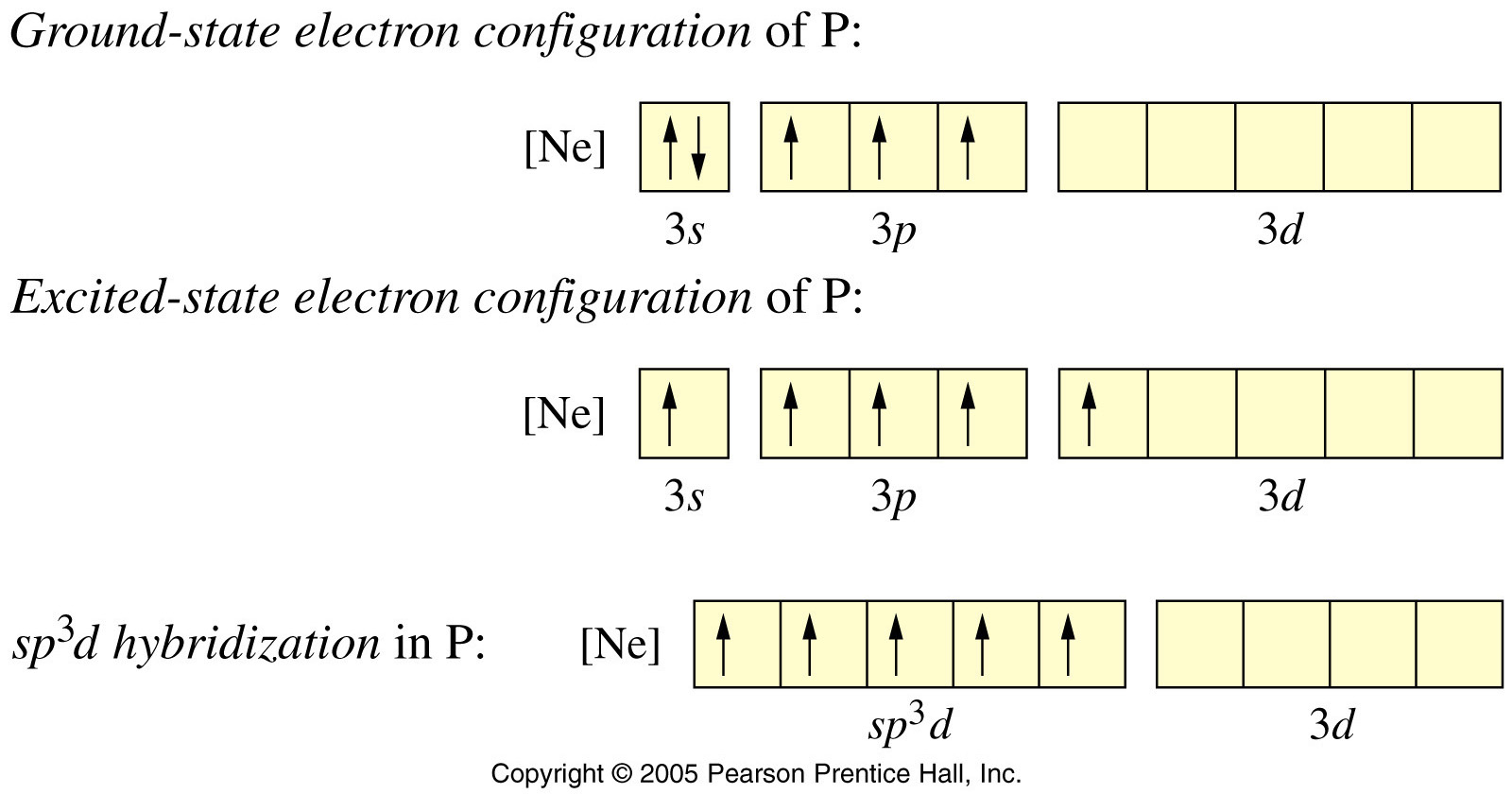
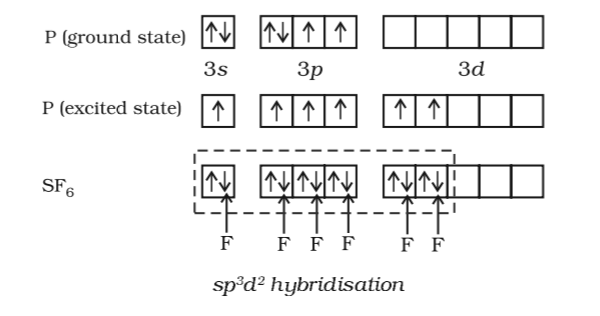
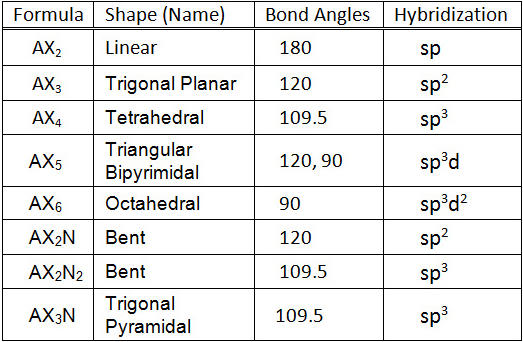
A molecule containing a central atom with sp3d2 hybridization has a(n) ______ electron geometry. a) trigonal planar b) trigonal bipyramidal c) octahedral d) tetrahedral e) trigonal pyramidal | Homework.Study.com

Sp3d2 Hybridization Has 1s 3p And 2d Orbitals That Undergo Intermixing To Form 6 Identical Sp3d2 Hybrid Orbitals Stok Vektör Sanatı & Kuantum'nin Daha Fazla Görseli - iStock

What type(s) of hybridization would you predict for molecules having the following general formula? AB_2U_4 | Homework.Study.com
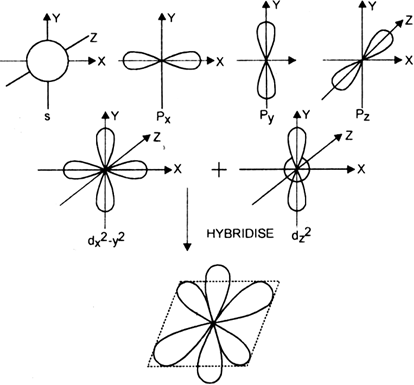
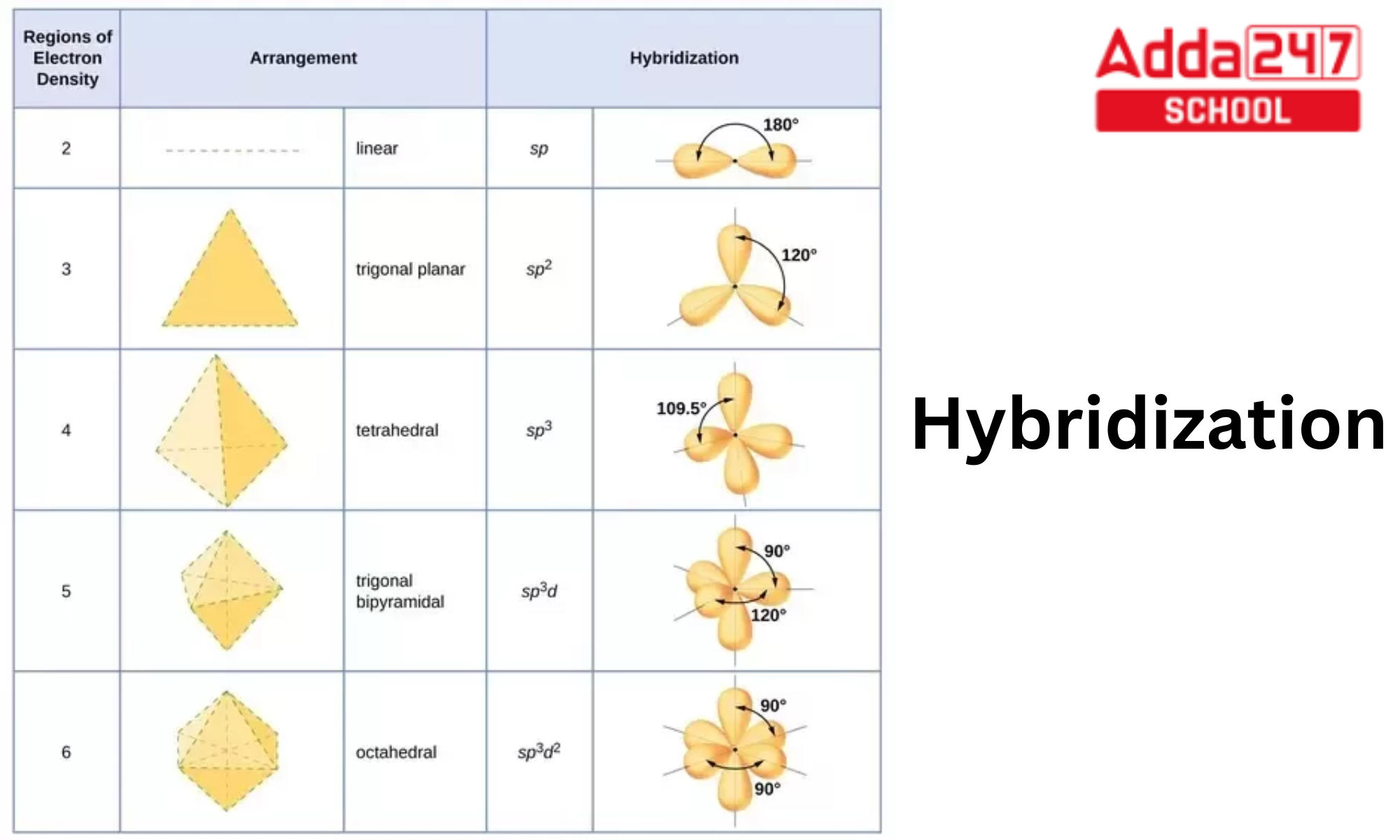
State and explain the geometric arrangements possible in sp3d and sp3d2 hybridisation. Name the d-orbitals involved in these. - Zigya


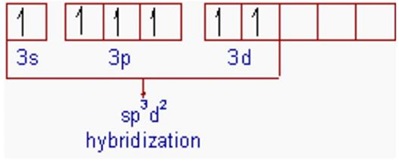
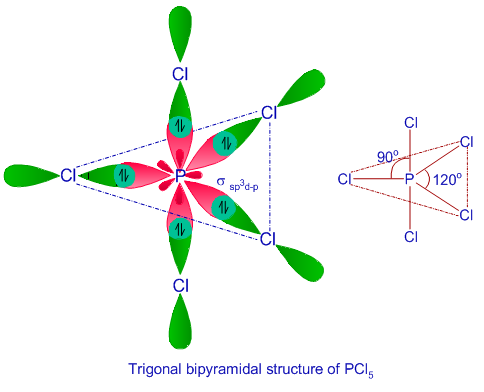
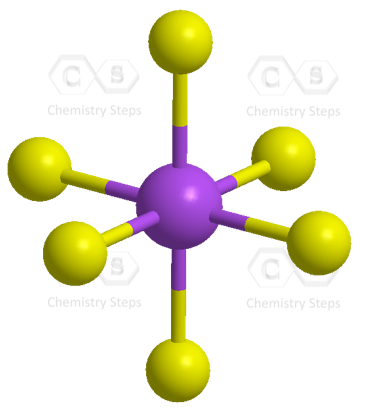





![sp3d2 hybridization is not displayed by : (1) BrF5 (2) SF6 (3) [CrF6]3− (4) PF5 - Brainly.in sp3d2 hybridization is not displayed by : (1) BrF5 (2) SF6 (3) [CrF6]3− (4) PF5 - Brainly.in](https://hi-static.z-dn.net/files/d39/27435abe12f9f9cf4f0688ed61384dea.jpg)

![Sp3d2 Hybridization - Download Free 3D model by AK Chemistry [53efdc3] - Sketchfab Sp3d2 Hybridization - Download Free 3D model by AK Chemistry [53efdc3] - Sketchfab](https://media.sketchfab.com/models/53efdc35286548f998c8e59f077c2a63/thumbnails/6f6b56c569b0494bbc41bf15c4c68b10/cf09f9e1b0b54997aff925cef1448918.jpeg)