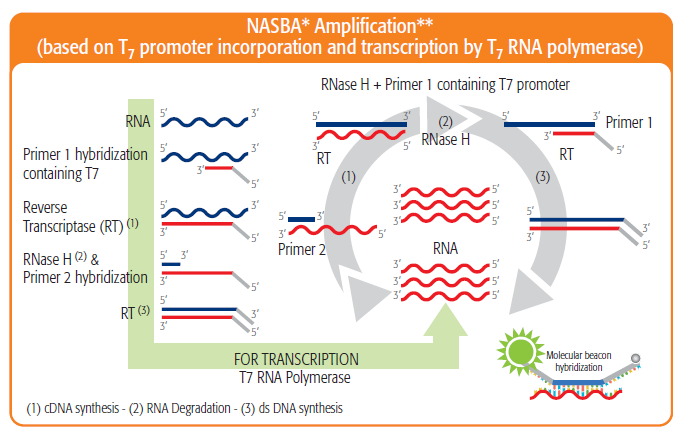
Sensitivity for HIV-1 antigen detection. Antigen detection sensitivity... | Download Scientific Diagram

PDF) Evaluation of the bioMérieux VIDAS HIV Duo Quick and Anti-HCV assays for dried blood spot based serosurveillance

Prolonged Second Diagnostic Window for Human Immunodeficiency Virus Type 1 in a Fourth-Generation Immunoassay: Are Alternative Testing Strategies Required? | Journal of Clinical Microbiology
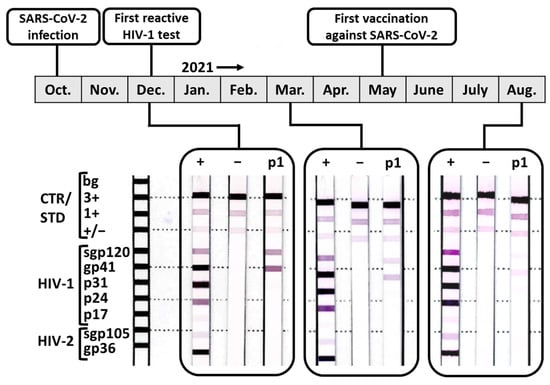
Viruses | Free Full-Text | False-Positive Screening and Confirmatory HIV Diagnostic Test in a Patient with Cured SARS-CoV-2 Infection Is Not Mediated by Env/Spike Cross-Reactive Antibodies

Utility Of POC Xpert HIV-1 Tests For Detection-Quantification Of Complex HIV Recombinants Using Dried Blood Spots From Kinshasa, D. R. Congo | Scientific Reports

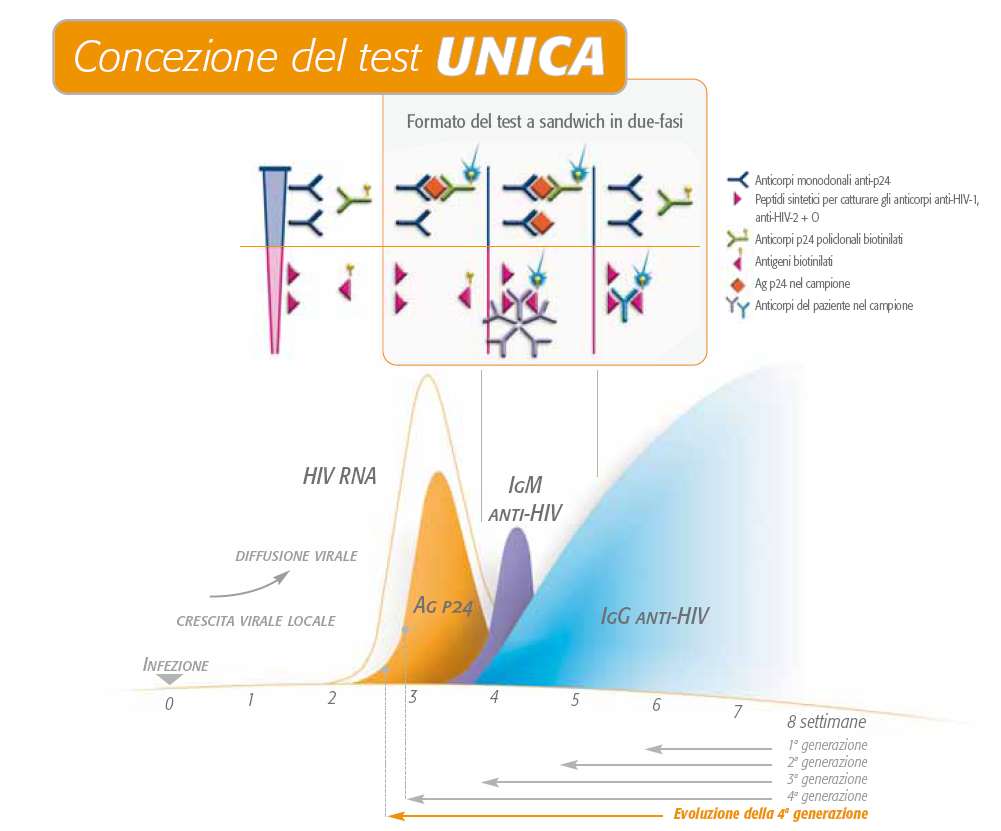
(PDF) Evaluation of a New Combined Antigen and Antibody Human Immunodeficiency Virus Screening Assay, VIDAS HIV DUO Ultra

LABORATORY DIAGNOSIS OF HIV INFECTION: ROLE OF COMBINED HIV P24 ANTIGEN AND ANTIBODY ASSAY | Semantic Scholar
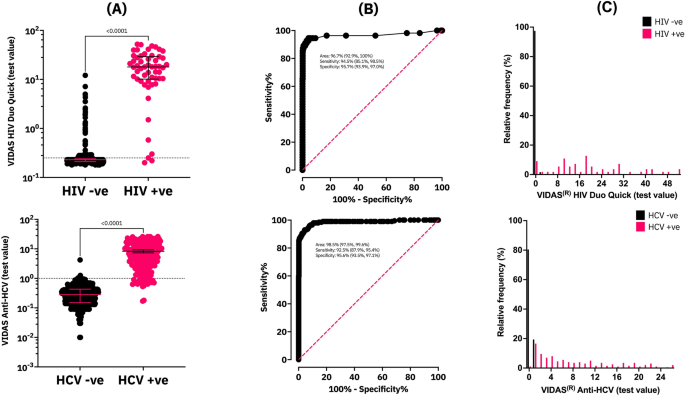
Evaluation of the bioMérieux VIDAS HIV Duo Quick and Anti-HCV assays for dried blood spot based serosurveillance | Scientific Reports
![PDF] A Human Immunodeficiency Virus Screening Algorithm to Address the High Rate of False-Positive Results in Pregnant Women in Japan | Semantic Scholar PDF] A Human Immunodeficiency Virus Screening Algorithm to Address the High Rate of False-Positive Results in Pregnant Women in Japan | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d792306d686814b37c8df9cc937f686f99ee2a22/3-Table1-1.png)